Solutions and Cells
Learning Targets:
1. Students can use the concept of concentration gradient to explain the movement of materials across the cell membrane.
2. Students can differentiate between the types of passive and active transport.
3. Students can describe the osmotic effects on a cell when placed in a hypotonic, hypertonic, or isotonic solution.
Key Vocabulary
- Concentration Gradient
- Hypertonic
- Hypotonic
- Isotonic
How do cells react in different solutions?
Understanding CONCENTRATION is the key.
Concentration is the amount of particles in water. Picture making Kool-Aid; if you make Kool-Aid in a glass with a full cup of sugar and only add enough water to fill it to the fop of the glass, that would be a HIGH CONCENTRATION Kool-Aid. If you make Kool-aid in a 5 gallon bucket with only a cup of sugar and fill the water to the top of the 5 gallon bucket, it will be a LOW CONCENTRATION of Kool-aid.
In class we will observe how concentration affects a cell using an egg, since we can't actually see cells!
In class we will observe how concentration affects a cell using an egg, since we can't actually see cells!
This movement of water to equal out concentration is called OSMOSIS!
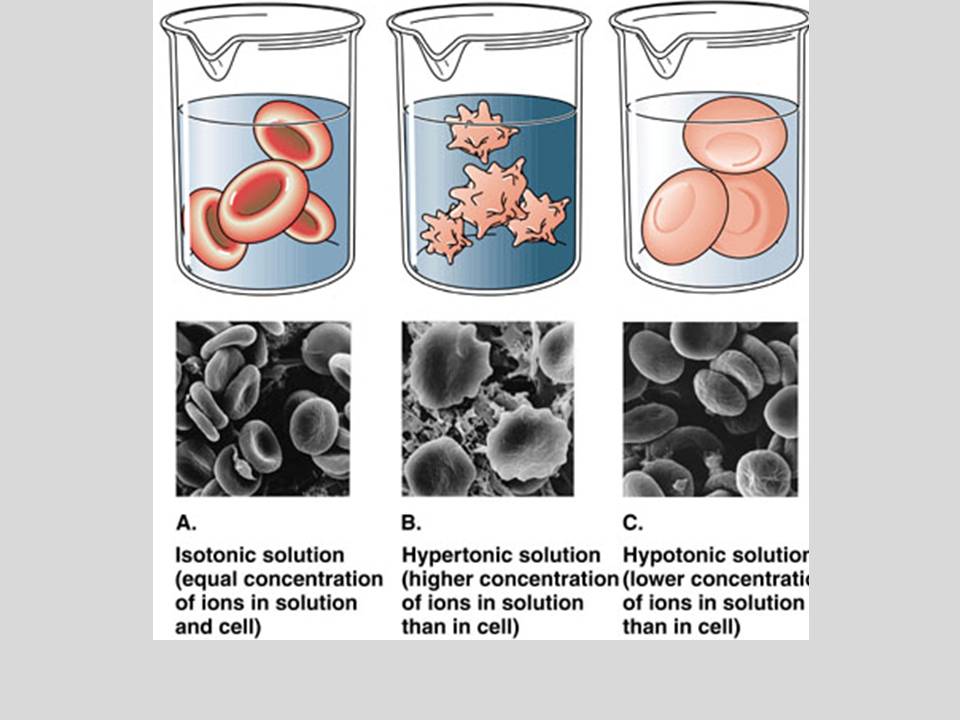
Isotonic SolutionsConcentration of the solution is EQUAL to the concentration inside the cell (or egg). This causes the cell (or egg) to STAY THE SAME SIZE!
|
Hypertonic SolutionsSolution's concentration is HIGHER than the concentrations inside the cell (or egg). This causes the cell (or egg) to SHRINK because the water from inside the cell (or egg) rushes out into the solution to even out the concentrations of both the solution and the cell. THE CELL GETS SMALLER CAUSING LOTS OF PROBLEMS FOR THE CELL!
|
Hypotonic SolutionsSolution's concentration is LOWER than the concentration inside the cell (or egg). This causes the cell (or egg) to EXPAND because the water from the solution in rushing inside to even out the concentrations. THE CELL GETS BIGGER CAUSING LOTS OF PROBLEMS FOR THE CELL!
|